About our research
Cell cycle checkpoints prevent a cell from dividing when they're damaged; instead, the damaged cell stops, repairs damage, and then continues its program. Drugs can affect DNA replication which slows and stops the cell in S-phase (S-phase and Intra-S checkpoints). DNA breaks and lesions formed before/during mitosis will also trigger a checkpoint (G2- and spindle-checkpoints). Specific checkpoint proteins are activated in response to the DNA damage, and this starts the checkpoint and maintains arrest. If a cell is missing a particular protein or checkpoint activity it cannot properly stop to repair; these "checkpoint mutant" cells usually do not survive drug exposure. Worse, some may survive but become mutated and different from the original population in the process!
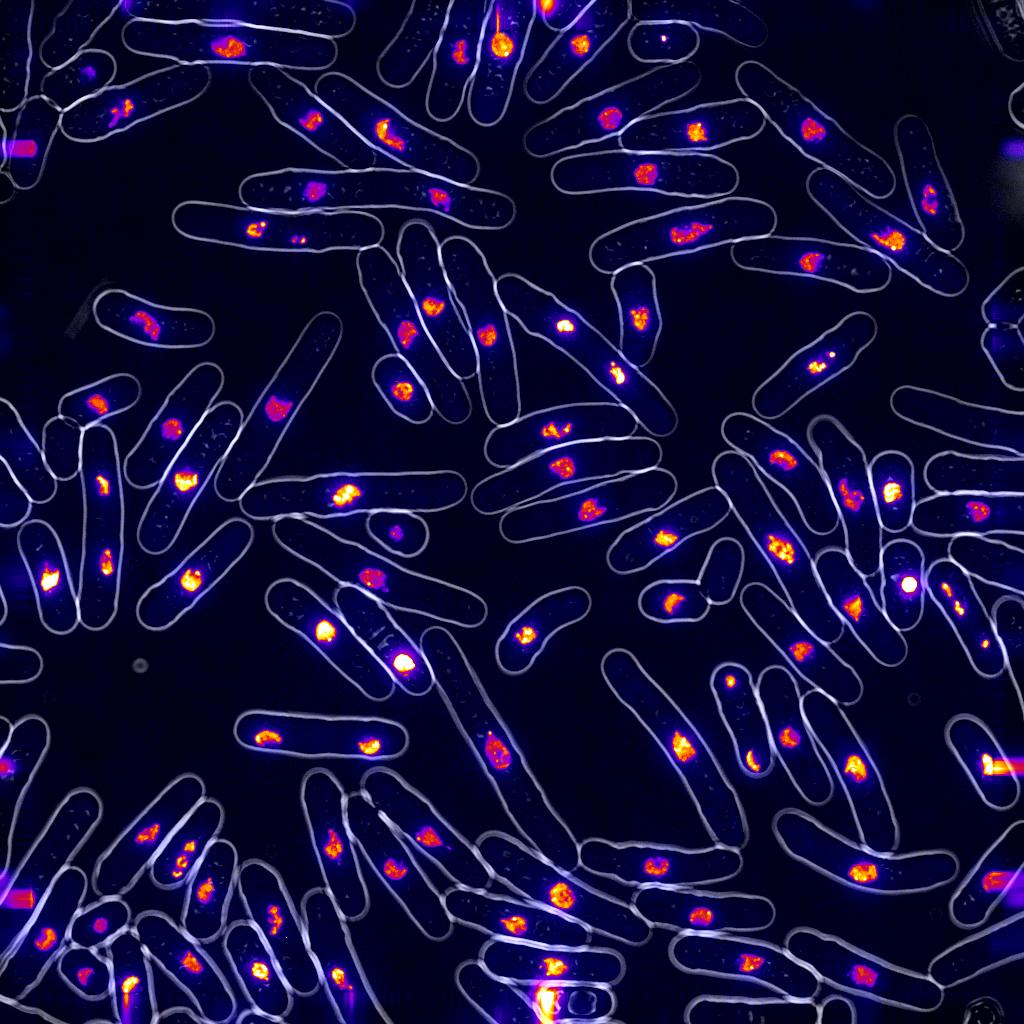 Environmental stress affects a cell's ability to trigger and/or maintain a checkpoint, but the mechanism and effects are not well known. Starving a cell for nutrients is one form of stress. Other kinds of environmental stress are temperature changes, pH, or osmolarity. We examine how specific environmental triggers can change survival during drug treatment. We also ask whether enhanced survival means more mutation.
Environmental stress affects a cell's ability to trigger and/or maintain a checkpoint, but the mechanism and effects are not well known. Starving a cell for nutrients is one form of stress. Other kinds of environmental stress are temperature changes, pH, or osmolarity. We examine how specific environmental triggers can change survival during drug treatment. We also ask whether enhanced survival means more mutation.
This is important, because ignoring - and then surviving- a checkpoint can have long-term consequences. We have previously found that cells which survive replication stress have higher mutation rates and chromosome rearrangements (ref - new paper). This is exciting, because conditions that create and promote DNA replication instability are well known contributors in carcinogenesis. After developing live-cell imaging methods for S.pombe, we can observe genome instability in real time and detect how environmental conditions/drugs interact.
What we want to find are the cells that survive the treatments. We call these the "Escapees", because of their circumstance. Considering that cancer cells frequently have checkpoint mutations, and that the environment for a cancer may be variable, we want to know whether Escapees are the cells that survive chemotherapy and cause patients to relapse later. We isolate Escapee populations and ask how they are different, why they survived when most cells died in drug. Using this knowledge, we target the mechanisms that allow them to survive, and determine two things :
1) how to prevent these cells from surviving under similar conditions. This is similar to a cancer cell that is being treated to ensure chemotherapy has a maximal effect.
2) ask whether the Escapees that survive are different from the original cells; do they have mutations?
Research funding for the Sabatinos Lab
